31. S. Dubey, S. Mandal, R. Sarkar, B. Koley, S. Roy, K. P. Bag, S. Rout, G. Sahoo, D. Bonne, and
T. Das* "Copper-Assisted Oxidative One-Pot Heterodifunctionalization of Cyclopentane-1,3-diones" The Journal of Organic Chemistry, 2026, 91, 1994−2003. (DOI: 10.1021/acs.joc.5c02411)
30. S. Roy, S. Maity, S. Dubey, K. P. Bag, M. Sau, M. Jana, S. Mahanta and T. Das *
"Methyl 3-(2-formyl-5-methoxynaphthalen-1-yl)propanoate as a fluorescent probe for folding and binding studies of human serum albumin" New Journal of Chemistry, 2025, DOI: 10.1039/D5NJ03358F (Advance Article).
.png)

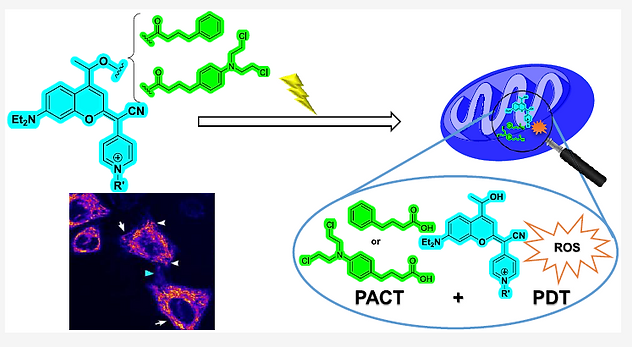
29. S. Dubey, P. Trivedi, M. Sau, S. Mandal, S. Roy, S. Rout, S. Das, G. Sahoo, D. Bonne and
T. Das * "Oxidative functionalization of cyclopentane-1,3-diones: cell imaging studies of diphenothiazine derivatives" Organic & Biomolecular Chemistry, 2025, 23, 8672 - 8677. (https://doi.org/10.1039/D5OB01082A)

28. S. Roy, T. K. Mukhopadhyay, M. Sau, S. Dubey, G. Gulfisha, K. Basudev, S. Nayak, G. Sahoo, S. Mahanta and T. Das* "Spectroscopic Behaviors of a Newly Synthesized Benzimidazole Probe (EBINP) with Native and Denatured Bovine Serum Albumin Supported by Docking and Molecular Dynamics" (New Journal of Chemistry, 2025, 49, 11767 DOI: https://doi.org/10.1039/D5NJ01026H)

27. S. Roy, D. Ghorai, A. Mukherjee, S. Kumar Mondal, S. Dubey, M. Sau, S. Ghosh,* and T. Das* "HaloindoleCarboxaldehyde as an Emerging Precursor for the Synthesis of Versatile Essential Skeletons" (ChemistrySelect, 2025, 10, e02427)
26. M. López-Corrale, E. Izquierdo-García, M. Bosch, T. Das, A. Llebaria, L. Josa-Culleré and V. Marchán* "Exploring the Phototherapeutic Applications of Mitochondria-Targeted COUPY Photocages of Antitumor Drugs" (Journal of Medicinal Chemistry, 2025, 2025, 68, 9, 9741-9754. DOI: 10.1021/acs.jmedchem.5c00550)
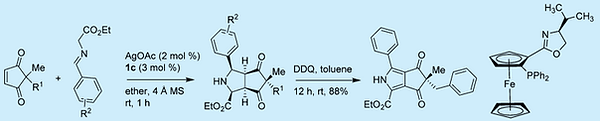
25. M. Sau, S. Dubey, S. Roy, J. Sahoo, G. Sahoo, N. Sepay, S. Chakraborty, S. Samanta, T. Das* "Pd/Cu dual catalysis for the oxidative dearomatization of pyrrole derivative: Mechanistic insights and biological studies." (European Journal of Organic Chemistry, 2024, accepted, DOI: 10.1002/ejoc.202400626)

24. S. Dubey, M. Sau, S. Roy, S. Mandal, G. Sahoo, D. Ghorai, A. Mukherjee, S. Mondal and T. Das* “Metal-Free C(sp2)-H Functionalization of Cyclopentene-1,3-dione: A Greener Approach for C-N and C-O Bond Formation Using Sodium Azide.” ChemistrySelect, 2024, 9, e202400747.

23. M. Sau, S. Dubey, S. Roy, S. Samanta, T. Das*, DBU Promoted Deformylative/DehalogenativeDifunctionalization to Access β-Bromovinyl AldehydesDerived from Tetralones. European Journal of Organic Chemistry 2023, e202300788.

22. S. Dubey , A. Pal , S. Roy , S. Sasmal , A. Tamrakar .R. Jana, T. Das* . "Recent advances on (3+ 2) cycloaddition of Azomethine Ylide" New Journal of Chemistry, 2023.
21. M. Sau, S. Dubey, J. Sahoo, G. Sahoo, P. Trivedi, A. Jana, S. Samanta, T. Das*, "Organocatalytic Approach for the Synthesis and Biological Studies of Naphthalene Fluorescent Probe through Hydrogen Transfer Reaction" European Journal of Chemistry, 2022, 46, e202201188.

20. S. Debnath, S. Parveen, P. Pradhan, I. Das, T. Das*, " Benzo[4,5]imidazo[1,2-a]pyridines and benzo[4,5]imidazo[1,2-a]pyrimidines: recent advancements in synthesis of two diversely important heterocyclic motifs and their derivatives" New Journal of Chemistry, 2022, 46, 10504-10534.
19. P. Pradhan, I. Das, S. Debnath, S. Parveen, T. Das*," Synthesis of Substituted Tropones and Advancement for the Construction of Structurally Significant Skeletons" Chemistry Select, 2022, 7, e202200440.
18. S. Thakur, A. Das, and T. Das* “1, 3 Dipolar Cycloaddition of Nitrones: Synthesis of Multisubstituted, Diverse Range of Heterocyclic Compounds” New Journal of Chemistry, 2021, 45, 11420-11456.

17. A. Das, S. Thakur, and T. Das* “Indole-2-Carboxaldehyde: An Emerging Precursor for the Construction of Diversified Imperative Skeleton” ChemistrySelect, 2021, 6, 4591–4619.

16. T. Das* “Desymmetrization of Cyclopentene-1,3-Diones via Alkylation, Arylation, Amidation and Cycloaddition Reactions”. Chemistry Select, 2020, 5, 14484-14509.

15. M. Sau, K. Verma, T. Das* “Synthesis of N-heterocycles via [4 + 3] cycloaddition of azomethine imine”. Journal of Heterocyclic Chemistry, 2020, 57, 3722-3734.
14. T. Das,* M. Sau, B. Daripa, D. Karmakar, S. Chakraborty. “[3+3] Cycloaddition of Azomethine Imine: Synthesis of Bior Tricyclic N-Heterocycle”. Chemistry Select, 2020, 5, 7605-7626.
13. S. K. Manna, T. Das, S. Samanta*. “Polycyclic Benzimidazole: Synthesis and Photophysical Properties”. Chemistry Select, 2019, 4, 8781-8790.
12. S. A. Ali, S. K. Mondal, T. Das, S. K. Manna, A. Bera, D. Dafadar, S., Naskar, M. R. Molla, S. Samanta. "One-pot tandem cyclisation to pyrrolo[1,2-a][1,4]-benzodiazepines: a modified approach to the Pictet–Spengler reaction". Organic & Biomolecular Chemistry, 2019, 17, 4652-4662.


11. J. Park, D-H. Kim, T. Das and C-G. Cho “Intramolecular Fischer indole synthesis for the direct synthesis of 3,4-fused tricyclic indole and application to the total synthesis of (-)- aurantioclavine.” Organic Letters, 2016, 18 (19), 5098-5101.

10. T. Das, A. Kayet, R. Mishra and V. K. Singh, “Highly Flourescent 1,2-Dihydropyrimido[1,6- α]indole: An efficient metal free Synthesis and Photophysical Study.” Chem. Commun., 2016, 52, 1.

9. T. Das, P. Saha and V. K. Singh*, “Silver(1)-Ferrophox Catalized Enantioselective Desymmetrization of Cyclopentenedione; Synthesis of Highly Substituted Bicyclic Pyrrolidines.” Organic Letters, 2015, 17 (20), 5088-5091.

8. R.K. Rej, T. Das, S. Hazra and S. Nanda*, “Chemoenzymatic Asymmetric Synthesis Fluoxetine, Atomoxetine, Nisoxetine, and Duloxetine.” Tetrahedron:Asymmetry, 2013, 24, 913-918.

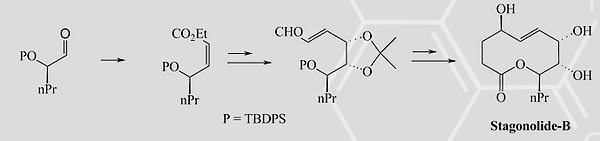
7. T. Das, T. Mahapatra and S. Nanda*, “ Total Synthesis of Stagonolide-B.” Tetrahedron Lett., 2012, 53, 1186-1189.
6. T. Das and S. Nanda* “Chemoenzymatic Total Synthesis of Stagonolide-E.” Tetrahedron Lett., 2012, 53, 256-258.

5. T. Mahapatra, T. Das and S. Nanda*, “Asymmetric Synthesis of Stagonolide-D and Stagonolide-G.” Bulletin of the Chemical Society of Japan, 2011, 84, 511-519.


4. T. Das, R. Bhuniya and S. Nanda*, “First Asymmetric Synthesis of Achaetolide.” Tetrahedron: Asymmetry, 2010, 21, 2206-2211.
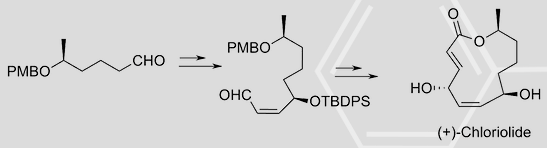
3. T. Das, N. Jana and S. Nanda*, “Asymmetric Synthesis of (+)-Chloriolide.” Tetrahedron Lett., 2010, 51, 2644-264.
2. R. Bhunya, N. Jana, T. Das and S. Nanda*, “Prunus Armenica Lyase (ParHNL) Catalyzed Asymmetric Synthesis of δ,ε-Unsaturated Cyanohydrins.” Synlett, 2009, 8, 1237-1240.

1. T. Mahapatra, T. Das and S. Nanda*, “Enantioselective Enzymatic Desymmetrizatio of Prochiral 1,3-diols and Enzymatic Resolution of Monoprotected 1,3-diols Based on α- tetralone and Related Multifunctional Scaffolds.” Tetrahedron: Asymmetry, 2008, 19, 2497- 2507.
